Acids
Acids are actually ionic compounds. There are two types of acids: those that contain oxygen, and those that don’t. Acids always have hydrogen in the front of the chemical formula, as is seen from these examples: HCl, HClO2, HF, and H2SO4.
For acids that do not contain oxygen, the name will always start with hydro-. With the second element, where you would change it to –ide in a binary compound, here it changes to –ic, and acid is also added. For example, HCl, we would have hydrochloric acid.
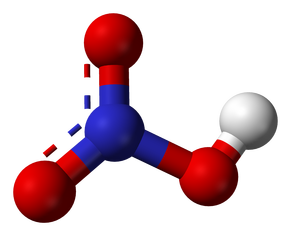
For acids that do contain oxygen, the naming depends on the polyatomic ion. If the polyatomic ion ends in –ate like nitrate, or NO3, it is changed to –ic as in nitric. In HNO3, there is no hydro- prefix, and acid is also added, so the end result is nitric acid.
But there are polyatomic ions that don’t end in –ate; usually they end in –ite. To name acids with these polyatomic ions, it is exactly the same as those that end in –ate, except the ending is –ous. For example, nitrite, or NO2, would change to nitrous, and when combined with hydrogen to make HNO2, it would be nitrous acid.
If you were to write the chemical formula of a given acid, you would most likely need a sheet with a list of basic polyatomic ions, as ion charges do matter and are very important.
Here is a quiz on naming acids: http://quizlet.com/662259/test/
You can change the types of questions and number questions presented in the test in the right sidebar.
HNO3, or nitric acid molecule
Apart from acids, there are also bases and salts. Bases are ionic compounds that end in OH, or hydroxide. Salts are ionic compounds that don't include hydrogen or hydroxide. To name bases and salts, it is very simple: just use the methods you would use to name binary ionic compounds or ionic compounds that include polyatomic ions. Examples of this include NaOH, which is sodium hydroxide (a base).